SERINC proteins
|
Initially some human SERINC genes were found to be overexpressed in some tumor-derived tissues and named TDE (tumour differentially expressed genes [1],[2]). However, the genes were later named SERINC (serine incorporator) based on some evidence that SERINCs favours the biosynthesis of serine-containing phospholipids [3]. An effect of SERINCs on the lipid composition of cellular membranes remains to be confirmed [4], [5].
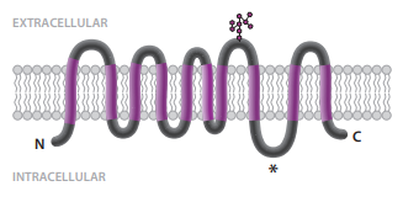
At the moment, the function of these proteins remains a puzzle, despite their wide distribution (from yeast to mammals, including plants). Eukaryotes encode one or more homologous multipass transmembrane proteins belonging to the SERINC family. Most orthologs are predicted to contain 10 transmembrane domains giving a similar topology with the N-terminus and the C-terminus in the cytoplasm. |
Schematic topology of the SERINC5 protein (modified from Firrito et al. Annual Review of Virology 2018. 5:323–40)
|
|
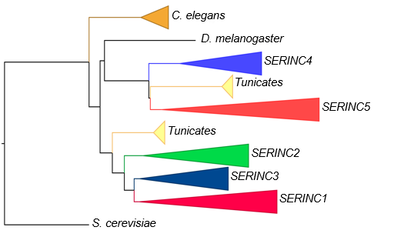
SERINC family of genes (modified from Firrito et al. Annual Review of Virology 2018. 5:323–40)
|
While only one SERINC gene is found in S. cerevisiae, C. elegans and D. melanogaster (named TMS1), higher eukaryotes have 5 homologous genes, resulting from two rounds of whole genome duplication events that occurred in vertebrates. SERINCs form two phylogenetically related groups, with human SERINC4 and 5 in one groups and SERINC1, 2, 3 in the other.
Different splicing isoforms are annotated in several animal genomes. In H. sapiens, some SERINC5 isoforms are predicted to have only 9 transmembrane domains, with an extracellular C-terminus [6]. It remains to be established whether these shorter proteins are functional. Nevertheless, the SERINC expansion during evolution was maintained in almost every vertebrate species and suggest a diversification of their function. |